All Disinfectants Are Not Created Equal
How to choose the right product for different needs that is both effective and skin friendly

Introduction
The COVID-19 pandemic has registered an exponential demand for hand disinfectants, given that unclean hands are one of the main sources of virus and disease transmission. Various products have entered the market to address the ongoing pandemic; however, consumers have sometimes mistakenly perceived some of these products as disinfectants or as having virucidal efficacy while they only have cosmetic-grade effectiveness.
This article is intended as a guide for Ecolab customers in the commercial and industrial sectors to select high-quality, effective and skin-friendly hand disinfectant products for their consumers and employees.
Overview of Disinfection and Disinfectant Products
Products with bactericidal, fungicidal, virucidal or any other efficacy against microorganisms, are identified as Biocides in Dubai. These products are regulated by special regulatory processes, which means they must first be evaluated and verified for effectiveness, safety and compliance by the Health and Safety department to then receive a national authorization from Dubai Municipality, before they are introduced to the market.
The reference legislation for biocide production in Dubai is the Technical Guidelines for Biocide (known as DM-PH&SD-GU82-BIO2) published on 14 April 2020 by Dubai Municipality.
Biocide Product Labels
Under the provisions of the Technical Guidelines for Biocides published by Dubai Municipality, commercial product labels must contain the necessary information to warn users of possible danger and the consequent steps to manage the associated risks of the misuse of products.
Below are the required components that must be declared in clear English and/or Arabic:
- Brand Name: a trademark or trade name.
- Product Name: a trademark or trade name cannot be used to replace the name of the biocidal product.
- Manufacturer details: name, address, web address of manufacturer or agent acting in UAE.
- Country of Origin: should be clearly indicated to prevent miscommunication with customers (e.g. USA, France, etc.) and not (NY, Paris, etc.).
- Ingredients: the identity of each active ingredient with its respective concentration in metric units.
- Size or weight of the product: must be clearly indicated on the product in metric units.
- Production and expiry date: must be clearly indicated on the label using non-removable printing or non-removable stickers.
- Storage Condition: to be clearly indicated mentioning any additional storage conditions beyond the normally required conditions.
- Usage Instructions: to be clearly indicated unless it is clear from the product name or presentation.
- Health Warning: to be clearly indicated according to the Globally Harmonized System (GHS).
- Batch Number: Batch or lot identification number to be indicated clearly on the label.
- Medical Claims: any claims related to prevention, treatment or cure of any disease type must not be presented or referred to on the labels, brochures, leaflets or advertisements or in any other way.
- Inconsistent illustrations: pictures and or illustrations which are inconsistent with the prevailing social customs and values shall not be used.
- Hazard symbols: should be used according to Globally Harmonized System (GHS).
Effectiveness of Biocide Products
Biocide products act against viruses, in addition to having bacterial and/or fungicide action. Most products on the market have an Ethyl Alcohol basis, the percentage of which is indicated on the label. There is a range of Alcohol concentration which is considered optimal for the product to be effective and this percentage is at least 70 percent of weight or higher. The concentration of Ethanol in a product can be indicated in volume or weight. If the percentage is indicated in volume of Alcohol per formulation volume (v/v), this will be less than the corresponding percentage indicated in weight/weight (w/w), because the density of Ethyl Alcohol is less than 1 g /cm3. For this reason, it is recommended to express concentration in a w/w formula because the weight factor, unlike volume, cannot be affected by temperature, specific weight, and concentration of reaction. It is important to verify that in the composition of the product, the indication of the presence of Ethyl Alcohol is indicated in weight and not in volume to ensure the correct concentration in the product.
Our Position at Ecolab
Ecolab has always been committed to ensuring public health through hygiene and disinfection of environments, operating in a wide range of institutional and industrial sectors, from healthcare, hospitality and catering, to industrial manufacturing processes in the food, cosmetics, automotive industries, working with three million customers in more than 170 countries around the world.
How products are manufactured is crucial to determining the quality that customers will ultimately receive. Our production facilities around the world are controlled by documented quality assurance systems to ensure the production processes and end results. The raw materials and end products are subject to strict quality controls, with specifications on the quality and quantity of the ingredients that are used in their make-up. The production of our formulations for the healthcare sector is based on Good Manufacturing Practices (GMP) standards and the manufacturing of products for other institutional and industrial sectors draws on the long experience and knowledge of hospital-grade standards for hygiene and disinfection.
Our products help clean and disinfect more than 40 billion hands every year to prevent the spread of infections and other diseases. Our hospital-grade products are used by thousands of healthcare facilities across the globe. All of our products have a high level of effectiveness and our hand disinfectants are superior to generic consumer products, as they offer excellent effectiveness in short application times.
Developed with the prospect of high-frequency use, our products are formulated with delicate ingredients, gentle on the skin that hydrate and protect hands, preventing the risk of allergies and sensitivities - which would make it problematic or impossible to continue sanitizing hands effectively. For instance, the presence of fragrance in a product increases the risk of allergy in a professional environment, where exposure to the product can be up to 20 times more frequent than in a non-professional environment.
Partnering with Ecolab
During the COVID-19 pandemic, Ecolab’s production was increased worldwide to meet the short and long-term needs of our customers in all sectors. Our personal hygiene solutions can complement your current package of products and services and will help further improve your operational efficiency by ensuring employee safety and health. Ecolab's strength and size offer a unique positioning to meet the needs of customers in any operating sector, with considerable experience in research and development, regulation and quality, which guarantees the delivery of high-quality products.
Conclusion
The choice of disinfectant products should consider its effectiveness as a basic factor and also offer guarantees on compatibility with the skin and a general simplicity and pleasantness of use. A product formulation that balances these elements effectively is the best way to achieve the goal of high compliance and standards. It is important for operators and consumers to be aware of product differences to be able to choose the best products available on the market.
Together we can work beyond the basic regulatory requirements to make education and awareness the essential pillars for a new hygiene culture. The importance of hand disinfection and the dissemination of high standards of compliance in facilities with regard to products, quality and effectiveness are fundamental to public well-being and safety. If we add ease of use, users’ acceptability and pleasantness on the skin, we realize how such additional elements can positively contribute to the creation of new and healthy habits for the well-being and safety of all.
Appendix:
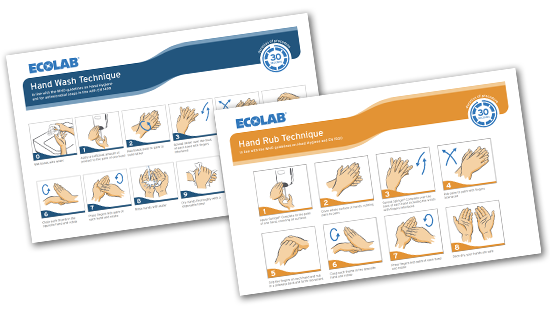
Hand Hygiene National and international institutions agree on the fundamental importance of careful and frequent hand hygiene through careful washing with soap and water, or with a hand disinfectant, if water is not available.
Sources:
- Dubai Municipality – Technical Guidelines for Biocides.
- Higher Institute of Health – Interim recommendations on disinfectants in the current COVID-19 emergency: medical-surgical and biocides. Release of April 25, 2020.



